The Patient's Pulse Oximeter Shows a Reading of 84
- Review
- Published:
Pulse oximetry
Critical Care volume nineteen, Article number:272 (2015) Cite this article
Abstract
Pulse oximetry is universally used for monitoring patients in the critical care setting. This article updates the review on pulse oximetry that was published in 1999 in Critical Care. A summary of the recently adult multiwavelength pulse oximeters and their ability in detecting dyshemoglobins is provided. The touch of the latest signal processing techniques and reflectance applied science on improving the performance of pulse oximeters during motion artifact and depression perfusion conditions is critically examined. Finally, data regarding the effect of pulse oximetry on patient outcome are discussed.
Introduction
Pulse oximetry is ubiquitously used for monitoring oxygenation in the disquisitional care setting. By forewarning the clinicians about the presence of hypoxemia, pulse oximeters may lead to a quicker treatment of serious hypoxemia and possibly circumvent serious complications. In this review, I update the principles of pulse oximetry from my article in 1999 and discuss recent technological advances that have been developed to enhance the accurateness and clinical applications of this monitoring technique [1]. Finally, available studies evaluating the impact of pulse oximetry on patient issue will besides be reviewed.
Principles of pulse oximetry
The technique of pulse oximetry has been previously described [1]. Using spectrophotometric methodology, pulse oximetry measures oxygen saturation past illuminating the skin and measuring changes in lite assimilation of oxygenated (oxyhemoglobin) and deoxygenated claret (reduced hemoglobin) using two lite wavelengths: 660 nm (red) and 940 nm (infrared) [1,2] (Fig. one). The ratio of absorbance at these wavelengths is calculated and calibrated against directly measurements of arterial oxygen saturation (SaO2) to establish the pulse oximeter's measure out of arterial saturation (SpO2). The waveform, which is available on most pulse oximeters, assists clinicians in distinguishing an antiquity from the truthful betoken (Fig. 2).
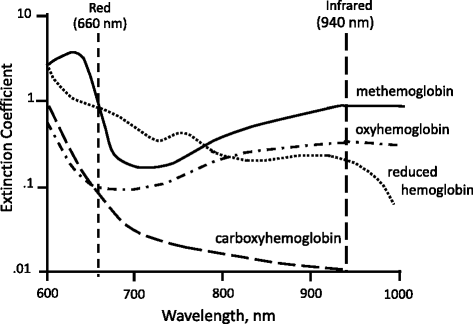
Transmitted light absorbance spectra of iv hemoglobin species: oxyhemoglobin, reduced hemoglobin, carboxyhemoglobin, and methemoglobin
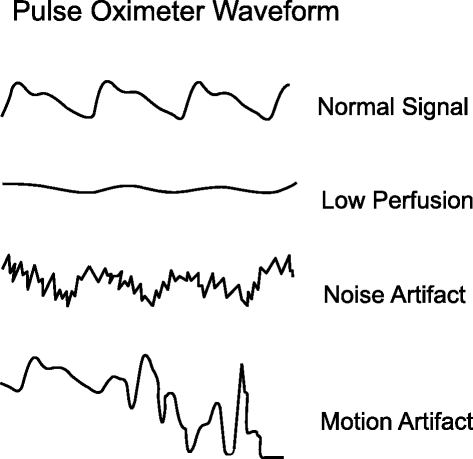
Mutual pulsatile signals on a pulse oximeter. (Pinnacle panel) Normal bespeak showing the sharp waveform with a clear dicrotic notch. (Second panel) Pulsatile signal during low perfusion showing a typical sine wave. (3rd panel) Pulsatile betoken with superimposed noise artifact giving a jagged appearance. (Bottom panel) Pulsatile betoken during motion antiquity showing an erratic waveform. Reprinted with permission from BioMed Central Ltd [1]
Accuracy
In critically ill patients with SaO2 values of 90 % or higher, the hateful difference between SpO2 and SaO2 (that is, bias) measured by a reference standard (CO-oximeter) is less than 2 %; the standard deviation of the differences between the 2 measurements (that is, precision) is less than 3 % [three–v]. The bias and precision of pulse oximetry readings, nonetheless, worsen when SaO2 is lower than ninety % [vi,7]. Although pulse oximetry is accurate in reflecting one-point measurements of SaOii, it does non reliably predict changes in SaOtwo, particularly in intensive intendance unit (ICU) patients [5,eight] (Fig. 3).

Changes in oxygen saturation measured by pulse oximetry (SpO2) compared with arterial oxygen saturation measured by a CO-oximeter (SaO2) in critically sick patients. The pulse oximeter consistently overestimated the bodily changes of SaOtwo. Reprinted with permission from BioMed Primal Ltd [eight]
The conventional pulse oximeters use transmission sensors in which the light emitter and detector are on opposing surfaces of the tissue bed. These sensors are suitable for use on the finger, toe, or earlobe; when tested under atmospheric condition of depression perfusion, finger probes performed improve than other probes [nine]. Recently, pulse oximeter probes that use reflectance applied science accept been developed for placement on the forehead [ten]. The reflectance sensor has emitter and detector components adjacent to 1 another, and then oxygen saturation is estimated from dorsum-scattered light rather than transmitted light. In critically ill patients with low perfusion, the bias and precision between SpO2 and SaO2 were lower for the forehead reflectance probe than for the finger probe [xi,12]. The superiority of forehead reflectance probes over conventional digital probes, withal, was non observed in patients with acute respiratory distress syndrome (ARDS) during a positive terminate-expiratory pressure (PEEP) recruitment maneuver [13].
The response fourth dimension of conventional oximeter probes varies; ear probes respond quicker to a change in Otwo saturation than finger probes [fourteen,fifteen]. A recent study compared the response time of the conventional finger probe with the reflectance forehead probe in patients undergoing general anesthesia [16] (Fig. 4). The lengths of time it took to detect a decrease in SpOii to 90 % after apnea was induced (desaturation response time) were 94 seconds for the forehead probe and 100 seconds for the finger probe. Later mask ventilation was started, the lengths of fourth dimension it took to detect an increase in SpO2 to 100 % (re-saturation response time) were 23.2 seconds for the brow probe and 28.9 seconds for the finger probes. The investigators speculated that the shorter response time with the reflectance forehead probe was about likely due to the location of the probe rather than to the workings of the reflectance engineering. The forehead probe monitors Oii saturation from the supraorbital avenue in which blood menses is abundant and is less likely to be affected by vasoconstriction than is a peripheral avenue [17].
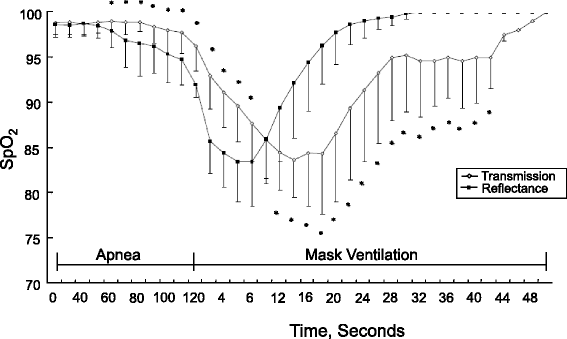
Oxygen saturation measured with pulse oximetry (SpO2) using transmittance finger probe (diamond) and reflectance forehead probe (squares) during apnea and mask ventilation with 100% Oii. The reflectance probe showed faster responses than the transmission probe at every measurement signal. *P < 0.05 between the two groups. Reprinted with permission from Wiley [16]
Limitations
Oximeters have limitations which may result in erroneous readings [15] (Table 1). Because of the sigmoid shape of the oxyhemoglobin dissociation curve, oximetry may not detect hypoxemia in patients with high arterial oxygen tension (PaOtwo) levels [i,18].
Conventional pulse oximeters can distinguish only two substances: reduced hemoglobin and oxyhemoglobin; it assumes that dyshemoglobins—such as carboxyhemoglobin (COHb) and methemoglobin (MetHb)—are absent (Fig. 1). Studies showed that the presence of elevated levels of COHb and MetHb could affect the accurateness of SpO2 readings [ane,19]. Accordingly, multiwavelength oximeters that are capable of estimating blood levels of COHb and MetHb accept recently been designed [xx]. In healthy volunteers, the accuracy of a multiwavelength oximeter (Masimo Rainbow-Prepare Rad-57 Pulse CO-oximeter; Masimo Corporation, Irvine, CA, USA) in measuring dyshemoglobins was evaluated by inducing carboxyhemoglobinemia (levels range from 0 % to 15 %) and methemoglobinemia (levels range from 0 % to 12 %) [20]. Bias between COHb levels measured with the pulse CO-oximeter and COHb levels measured with the laboratory CO-oximeter (standard method) was −1.22 %; the corresponding precision was 2.19 %. Bias ± precision of MetHB measured with the pulse CO-oximeter and MetHb measured with the laboratory CO-oximeter was 0.0 % ± 0.45 %. The accuracy of pulse CO-oximeters in measuring COHb levels was besides assessed during hypoxia [21]. In 12 good for you volunteers, the pulse CO-oximeter was authentic in measuring COHb at an SaO2 of less than 95 % (bias of −0.7 % and precision of 4.0 %); however, when the SaOtwo dropped below 85%, the pulse CO-oximeter was unable to measure COHb levels. In patients evaluated in the emergency section with suspected carbon monoxide poisoning, the bias between pulse CO-oximetric measurement of COHb and laboratory CO-oximetric measurement of COHb was less than iii % [22,23]. The limits of understanding betwixt the measurements, however, were large (−11.6 % to 14.14 %) [23], leading some authors to conclude that these new pulse CO-oximeters may not be used interchangeably with standard laboratory measurements of COHb [22–24].
Inaccurate readings with pulse oximetry have been reported with intravenous dyes used for diagnostic purposes, depression perfusion states (that is, depression cardiac output, vasoconstriction, and hypothermia), pigmented subjects and in patients with sickle cell anemia [ane,6,25,26]. Because the two wavelengths (660 and 940 nm) that pulse oximeters use to measure SpO2 can exist produced past various ambient light sources, the presence of such sources could produce imitation SpO2 readings. To examination the accurateness of pulse oximetry in the presence of ambient lite, Fluck and colleagues [27] performed a randomized controlled trial in good for you subjects in which SpO2 measurements were obtained in a photographic darkroom under 5 separate light sources: quartz-halogen, infrared, incandescent, fluorescent, and bilirubin light [27]. The largest departure in SpO2 between the control condition (that is, consummate darkness) and any of the 5 light sources was less than 5%. Smash polish can interfere with pulse oximetry readings [28]. In 50 critically ill patients requiring mechanical ventilation, Hinkelbein and colleagues [29] found that the mean difference betwixt SpOii and SaOii was greatest for blackness (+one.vi % ± 3.0 %), purple (+1.two % ± ii.6 %), and night blue (+1.i % ± 3.5 %) blast shine; limits of agreement ranged from half-dozen % (unpainted fingernail) to 14.four % (nighttime blue) (Fig. v). Rotating the oximeter finger probe past xc ° did non eliminate the error induced with nail polish.
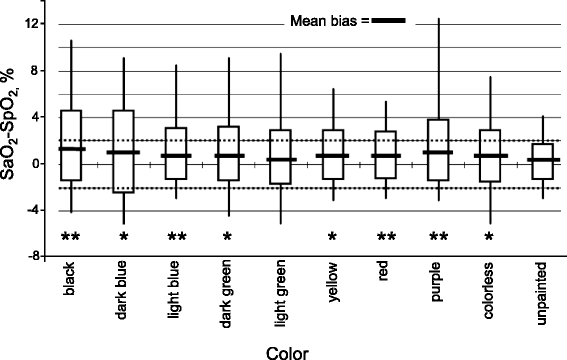
Bias of Otwo saturation pulse oximetry (SpO2) and arterial O2 saturation (SaOtwo) of various nail smooth colors in critically ill patients. Thick horizontal lines represent mean bias, the whiskers correspond maximum and minimum bias; the bottom and tiptop of the boxes represent the first and third quartiles. *P < 0.05 ,**P < 0.01 when compared with arterial oxygen saturation. Reprinted with permission from Elsevier Inc. [29]
Motion artifact is considered an of import crusade of error and fake alarms [thirty–33]. In the 1990s, several bespeak processing techniques were incorporated in pulse oximeters in an attempt to reduce motion antiquity [34–38]. One such technique is Masimo signal extraction applied science (SET™) [39]. During movement and hypoxia, the Masimo Fix oximeter performed better than the Agilent Viridia 24C (Agilent Technologies, Santa Clara, CA, Us), the Datex-Ohmeda 3740 (Datex-Ohmeda, Madison, WI, Usa), and the Nellcor North-395 (Covidien Corporation, Dublin, Ireland) oximeters [34].
The knowledge about pulse oximetry among clinicians continues to be limited. When 551 disquisitional care nurses were recently interviewed, 37 % of them did non know that oximeters were more likely to be inaccurate during patient motion, 15 % did not know that poor signal quality can produce inaccurate readings, and 30 % considered that SpOtwo readings could exist used in lieu of arterial blood gas samples when managing ICU patients [40].
Clinical applications
Pulse oximetry tin provide an early warning of hypoxemia [41,42]. In the largest randomized trial involving more than 20,000 perioperative patients, rates of incidence of hypoxemia (SpO2 of less than 90 %) were 7.9 % in patients who were monitored with pulse oximetry and only 0.4 % in patients without an oximeter [43]. The anesthesiologists reported that oximetry led to a alter in therapy on at least 1 occasion in up to 17 % of the patients. Using 95,407 electronically recorded pulse oximetry measurements from patients who underwent non-cardiac surgery at two hospitals, Ehrenfeld and colleagues [44] reported that during the intraoperative period, 6.8 % of patients had a hypoxemic event (SpO2 of less than 90) and 3.five % of patients had a severe hypoxemic event (SpO2 of non more than 85 %) lasting more 2 minutes. Hypoxemic events occurred generally during the induction or emergent stage of anesthesia; these fourth dimension periods are consistent with the clinical view that anesthesia-transitional states are high-run a risk periods for hypoxemia [45]. In patients undergoing gastric featherbed surgery, continuous monitoring of SpO2 revealed that episodic hypoxemia (SpO2 of less than 90 % for at least xxx seconds) occurred in all patients. For each patient, desaturation lasted equally long as 21 ± 15 minutes [46].
Pulse oximetry has been shown to be reliable in titrating the fractional inspired oxygen concentration (FIO2) in patients requiring mechanical ventilation; aiming for an SpOtwo of 92 % is reasonable for ensuring satisfactory oxygenation in Caucasian patients [6]. To decide whether the ratio of SpO2 to FIOtwo (S/F) can be used as a surrogate for the ratio of PaO2 to FIOii (P/F), SpO2 and PaO2 data from 1,074 patients with acute lung injury or ARDS who were enrolled in two large clinical trials were compared [47]. An S/F ratio of 235 predicted a P/F ratio of 200 (oxygenation benchmark for ARDS), a sensitivity of 0.85, and a specificity of 0.85. An S/F ratio of 310 reflected a P/F ratio of 300 (oxygenation criterion for acute lung injury), a sensitivity of 0.91, and a specificity of 0.56. In patients undergoing surgery, the S/F ratio was shown to be a reliable proxy for the P/F ratio (correlation coefficient (r) of 0.46), specially in those patients requiring PEEP levels of greater than ix cm H2O (r = 0.68) and those patients with a P/F ratio of 300 or less (r = 0.61) [48]. In the ICU, the S/F ratio tin can also exist a surrogate measure for the P/F ratio when calculating the sequential organ failure assessment score, which measures the severity of organ dysfunction in critically sick patients [49].
Cost-effectiveness
Studies accept shown that the presence of pulse oximetry may reduce the number of arterial claret gas samples obtained in the ICU and in the emergency department [l,51]. Still, the lack of incorporating explicit guidelines for the advisable employ of pulse oximetry may lessen the toll-effectiveness of pulse oximetry in the ICU [1].
Effect on outcome
To engagement, the largest randomized controlled trial that has evaluated the impact of pulse oximetry on outcome was the report by Moller and colleagues [43] in xx,802 surgical patients. Although myocardial ischemia occurred less oftentimes in the oximetry than the control group, the numbers of post-operative complications and hospital deaths were similar in the 2 groups [43].
In a more contempo randomized study in ane,219 postal service-operative patients, Ochroch and colleagues [52] assessed the impact of pulse oximetry on the charge per unit of transfer to the ICU from a post-surgical care floor. Upon access to the report flooring, patients were randomly assigned to receive monitoring with a pulse oximeter either continuously (northward = 589) (oximeter group) or intermittently (n = 630) according to clinical needs as judged by a nurse or a doc (control group). The percentages of patients transferred to the ICU were like in the oximeter group and the command group (6.7 % versus 8.5 %). A lower rate of ICU transfers for pulmonary complications was noted in the oximeter group. For those patients who required ICU transfer, the estimated cost from enrollment to completion of the written report was less in the oximeter grouping ($15,481) than in the control group ($18,713) despite the older age and higher comorbidity of the former. The authors speculate that reduction in pulmonary transfers to the ICU may be due to the earlier recognition and handling of postal service-operative pulmonary complications.
The lack of demonstrable benefit of pulse oximetry on outcome in clinical trials may be due to the signal-to-racket ratio [41,53]. Because the outcome under evaluation (readmission to the ICU, myocardial infarction, or decease) is rare, a huge number of patients are needed to evidence a reduction in these events [41]. To demonstrate a reduction in complications in the study by Moller and colleagues, for case, a 23-fold increase in enrollment would have been required [41,53].
The fact that randomized trials failed to demonstrate that routine monitoring with pulse oximetry improved patient outcome has non stopped anesthesiologists from using pulse oximeters [53,54]. When surveyed, 94 % of the anesthesiologists in the study past Moller and colleagues [43] considered the pulse oximeters to exist helpful in guiding clinical direction. They believed that maintaining oxygenation inside the physiologic limits with the assist of pulse oximetry might assist avoid irreversible injury. Information technology is this perspective that has made pulse oximetry a crucial role of standard of care despite the absence of proven efficacy [41].
Conclusions
Pulse oximetry is universally used for monitoring respiratory status of patients in the ICU. Recent advances in signal analysis and reflectance engineering have improved the performance of pulse oximeters under conditions of motility artifact and low perfusion. Multiwavelength oximeters may show to be useful in detecting dyshemoglobinemia. Monitoring with pulse oximetry continues to exist a critical component of standard of intendance of critically sick patients despite the paucity of information that such devices ameliorate outcome.
Abbreviations
- ARDS:
-
acute respiratory distress syndrome
- COHb:
-
carboxyhemoglobin
- FIO2 :
-
fractional inspired oxygen concentration
- ICU:
-
intensive intendance unit
- MetHb:
-
methemoglobin
- PaO2 :
-
arterial oxygen tension
- PEEP:
-
positive finish-expiratory pressure
- P/F:
-
PaOii-to-FIO2 ratio
- r:
-
correlation coefficient
- SaO2 :
-
arterial oxygen saturation
- SET:
-
signal extraction technology
- S/F:
-
SpO2-to-FIO2 ratio
- SpO2 :
-
oxygen saturation measured past pulse oximetry
References
-
Jubran A. Pulse oximetry. Crit Intendance. 1999;3:R11–vii.
-
Wukitsch MW, Petterson MT, Tobler DR, Pologe JA. Pulse oximetry: assay of theory, technology, and exercise. J Clin Monit. 1988;4:290–301.
-
Wouters PF, Gehring H, Meyfroidt Thou, Ponz L, Gil-Rodriguez J, Hornberger C, et al. Accuracy of pulse oximeters: the European multi-center trial. Anesth Analg. 2002;94(i Suppl):S13–vi.
-
Webb RK, Ralston Air conditioning, Runciman WB. Potential errors in pulse oximetry, II. Effects of changes in saturation and signal quality. Amazement. 1991;46:207–12.
-
Van de Louw A, Cracco C, Cerf C, Harf A, Duvaldestin P, Lemaire F, et al. Accuracy of pulse oximetry in the intensive intendance unit. Intensive Care Med. 2001;27:1606–13.
-
Jubran A, Tobin MJ. Reliability of pulse oximetry in titrating supplemental oxygen therapy in ventilator-dependent patients. Chest. 1990;97:1420–v.
-
Pulse oximeters. Health Devices. 1989;eighteen:185–99, 206–19, 222–thirty.
-
Perkins GD, McAuley DF, Giles Southward, Routledge H, Gao F. Do changes in pulse oximeter oxygen saturation predict equivalent changes in arterial oxygen saturation? Crit Care. 2003;seven:R67.
-
Clayton DG, Webb RK, Ralston AC, Duthie D, Runciman WB. Pulse oximeter probes. A comparison betwixt finger, nose, ear and forehead probes under conditions of poor perfusion. Amazement. 1991;46:260–5.
-
Branson RD, Mannheimer PD. Brow oximetry in critically ill patients: the example for a new monitoring site. Respir Care Clin N Am. 2004;x:359–67.
-
Fernandez M, Burns K, Calhoun B, George S, Martin B, Weaver C. Evaluation of a new pulse oximeter sensor. Am J Crit Care. 2007;16:146–52.
-
Schallom Fifty, Sona C, McSweeney Grand, Mazuski J. Comparing of forehead and digit oximetry in surgical/trauma patients at risk for decreased peripheral perfusion. Eye Lung. 2007;36:188–94.
-
Hodgson CL, Tuxen DV, Holland AE, Keating JL. Comparison of brow Max-Fast pulse oximetry sensor with finger sensor at loftier positive cease-expiratory pressure in adult patients with acute respiratory distress syndrome. Anaesth Intensive Care. 2009;37:953–60.
-
Immature D, Jewkes C, Spittal Grand, Blogg C, Weissman J, Gradwell D. Response time of pulse oximeters assessed using astute decompression. Anesth Analg. 1992;74:189–95.
-
Jubran A. Pulse oximetry. In: Tobin MJ, editor. Principles and Practice of Intensive Care Monitoring. New York: McGraw-Colina, Inc; 1998. p. 261–87.
-
Choi SJ, Ahn HJ, Yang MK, Kim CS, Sim WS, Kim JA, et al. Comparing of desaturation and resaturation response times between transmission and reflectance pulse oximeters. Acta Anaesthesiol Scand. 2010;54:212–seven.
-
MacLeod DB, Cortinez LI, Keifer JC, Cameron D, Wright DR, White WD, et al. The desaturation response time of finger pulse oximeters during mild hypothermia. Anaesthesia. 2005;sixty:65–71.
-
Ralston Air conditioning, Webb RK, Runciman WB. Potential errors in pulse oximetry, I. Pulse oximeter evaluation. Amazement. 1991;46:202–vi.
-
Buckley RG, Aks SE, Eshom JL, Rydman R, Schaider J, Shayne P. The pulse oximetry gap in carbon monoxide intoxication. Ann Emerg Med. 1994;24:252–5.
-
Barker SJ, Back-scratch J, Redford D, Morgan S. Measurement of carboxyhemoglobin and methemoglobin by pulse oximetry: a human volunteer study. Anesthesiology. 2006;105:892–7.
-
Feiner JR, Rollins MD, Sall JW, Eilers H, Au P, Bickler PE. Accurateness of carboxyhemoglobin detection by pulse CO-oximetry during hypoxemia. Anesth Analg. 2013;117:847–58.
-
Sebbane G, Blood PG, Mercier M, Lefebvre S, Thery R, Dumont R, et al. Emergency department direction of suspected carbon monoxide poisoning: role of pulse CO-oximetry. Respir Care. 2013;58:1614–20.
-
Touger M, Birnbaum A, Wang J, Chou Chiliad, Pearson D, Bijur P. Operation of the RAD-57 pulse CO-oximeter compared with standard laboratory carboxyhemoglobin measurement. Ann Emerg Med. 2010;56:382–8.
-
Maisel WH, Lewis RJ. Noninvasive measurement of carboxyhemoglobin: how authentic is accurate plenty? Ann Emerg Med. 2010;56:389–91.
-
Saito S, Fukura H, Shimada H, Fujita T. Prolonged interference of bluish dye 'patent blue' with pulse oximetry readings. Acta Anaesthesiol Scand. 1995;39:268–9.
-
Comber JT, Lopez BL. Evaluation of pulse oximetry in sickle cell anemia patients presenting to the emergency department in acute vasoocclusive crisis. Am J Emerg Med. 1996;14:16–8.
-
Fluck Jr RR, Schroeder C, Frani G, Kropf B, Engbretson B. Does ambient lite affect the accuracy of pulse oximetry? Respir Care. 2003;48:677–80.
-
Cote CJ, Goldstein EA, Fuchsman WH, Hoaglin DC. The consequence of nail smooth on pulse oximetry. Anesth Analg. 1988;67:683–half dozen.
-
Hinkelbein J, Genzwuerker HV, Sogl R, Fiedler F. Effect of nail smoothen on oxygen saturation determined by pulse oximetry in critically ill patients. Resuscitation. 2007;72:82–91.
-
Reich DL, Timcenko A, Bodian CA, Kraidin J, Hofman J, DePerio M, et al. Predictors of pulse oximetry data failure. Anesthesiology. 1996;84:859–64.
-
Moller JT, Pedersen T, Rasmussen LS, Jensen PF, Pedersen BD, Ravlo O, et al. Randomized evaluation of pulse oximetry in 20,802 patients: I. Design, demography, pulse oximetry failure rate, and overall complication charge per unit. Anesthesiology. 1993;78:436–44.
-
Runciman WB, Webb RK, Barker L, Currie M. The Australian Incident Monitoring Study. The pulse oximeter: applications and limitations - an analysis of 2000 incident reports. Anaesth Intensive Care. 1993;21:543–50.
-
Rheineck-Leyssius AT, Kalkman CJ. Influence of pulse oximeter lower alarm limit on the incidence of hypoxaemia in the recovery room. Br J Anaesth. 1997;79:460–4.
-
Barker SJ. 'Motion-resistant' pulse oximetry: a comparison of new and sometime models. Anesth Analg. 2002;95:967–72.
-
Petterson MT, Begnoche VL, Graybeal JM. The consequence of motion on pulse oximetry and its clinical significance. Anesth Analg. 2007;105(6 Suppl):S78–84.
-
Next-generation pulse oximetry. Health Devices. 2003;32:49–103.
-
Barker SJ, Shah NK. The furnishings of motion on the functioning of pulse oximeters in volunteers (revised publication). Anesthesiology. 1997;86:101–8.
-
Dumas C, Wahr JA, Tremper KK. Clinical evaluation of a prototype motility artifact resistant pulse oximeter in the recovery room. Anesth Analg. 1996;83:269–72.
-
Pollard V, Prough DS. Signal extraction applied science: a improve mousetrap? Anesth Analg. 1996;83:213–4.
-
Giuliano KK, Liu LM. Knowledge of pulse oximetry among critical care nurses. Dimens Crit Care Nurs. 2006;25:44–9.
-
Jubran A, Tobin MJ. Monitoring during mechanical ventilation. In: Tobin MJ, editor. Principles and Practice of Mechanical Ventilation. New York: McGraw-Hill, Inc; 2013. p. 261–87.
-
Pretto JJ, Roebuck T, Beckert Fifty, Hamilton G. Clinical employ of pulse oximetry: official guidelines from the Thoracic Society of Australia and New Zealand. Respirology. 2014;19:38–46.
-
Moller JT, Johannessen NW, Espersen One thousand, Ravlo O, Pedersen BD, Jensen PF, et al. Randomized evaluation of pulse oximetry in twenty,802 patients: II. Perioperative events and postoperative complications. Anesthesiology. 1993;78:445–53.
-
Ehrenfeld JM, Funk LM, Van Schalkwyk J, Merry AF, Sandberg WS, Gawande A. The incidence of hypoxemia during surgery: prove from 2 institutions. Can J Anaesth. 2010;57:888–97.
-
Hare GM, Kavanagh BP. Hypoxemia during surgery: learning from history, science, and current exercise. Can J Anaesth. 2010;57:877–81.
-
Gallagher SF, Haines KL, Osterlund LG, Mullen Yard, Downs JB. Postoperative hypoxemia: common, undetected, and unsuspected after bariatric surgery. J Surg Res. 2010;159:622–6.
-
Rice TW, Wheeler AP, Bernard GR, Hayden DL, Schoenfeld DA, Ware LB, et al. Comparing of the SpO2/FIO2 ratio and the PaO2/FIO2 ratio in patients with acute lung injury or ARDS. Chest. 2007;132:410–7.
-
Tripathi RS, Blum JM, Rosenberg AL, Tremper KK. Pulse oximetry saturation to fraction inspired oxygen ratio as a measure of hypoxia under general anesthesia and the influence of positive end-expiratory pressure level. J Crit Intendance. 2010;25:542.e9-13.
-
Pandharipande PP, Shintani AK, Hagerman HE, St Jacques PJ, Rice TW, Sanders NW, et al. Derivation and validation of Spo2/Fio2 ratio to impute for Pao2/Fio2 ratio in the respiratory component of the Sequential Organ Failure Assessment score. Crit Intendance Med. 2009;37:1317–21.
-
Le Bourdelles G, Estagnasie P, Lenoir F, Brun P, Dreyfuss D. Use of a pulse oximeter in an adult emergency department: bear on on the number of arterial claret gas analyses ordered. Chest. 1998;113:1042–7.
-
Solsona JF, Marrugat J, Vazquez A, Masdeu G, Alvarez F, Nolla J. Consequence of pulse oximetry on clinical practice in the intensive care unit. Lancet. 1993;342:311–2.
-
Ochroch EA, Russell MW, Hanson third WC, Devine GA, Cucchiara AJ, Weiner MG, et al. The bear on of continuous pulse oximetry monitoring on intensive care unit admissions from a postsurgical care floor. Anesth Analg. 2006;102:868–75.
-
Shah A, Shelley KH. Is pulse oximetry an essential tool or just another distraction? The role of the pulse oximeter in modern anesthesia care. J Clin Monit Comput. 2013;27:235–42.
-
Pedersen T, Nicholson A, Hovhannisyan G, Moller AM, Smith AF, Lewis SR. Pulse oximetry for perioperative monitoring. Cochrane Database Syst Rev. 2014;three, CD002013.
Author information
Affiliations
Respective author
Additional data
Competing interests
The author declares that he has no competing interests.
Come across related review past Jubran, http://ccforum.com/content/three/2/R11
This is an Open Admission article distributed nether the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/past/4.0), which permits unrestricted employ, distribution, and reproduction in whatsoever medium, provided the original work is properly cited. The Creative Eatables Public Domain Dedication waiver (http://creativecommons.org/publicdomain/goose egg/1.0/) applies to the data made available in this commodity, unless otherwise stated.
Rights and permissions
About this article
Cite this article
Jubran, A. Pulse oximetry. Crit Care 19, 272 (2015). https://doi.org/x.1186/s13054-015-0984-eight
-
Published:
-
DOI : https://doi.org/10.1186/s13054-015-0984-8
Keywords
- Pulse Oximetry
- Pulse Oximeter
- Sequential Organ Failure Assessment Score
- COHb Level
- Finger Probe
Source: https://ccforum.biomedcentral.com/articles/10.1186/s13054-015-0984-8
0 Response to "The Patient's Pulse Oximeter Shows a Reading of 84"
Postar um comentário